Department of Cancer Biology
Our Research Focus

Signaling Mechanisms Controlling Tumor Growth and Immune System Interactions
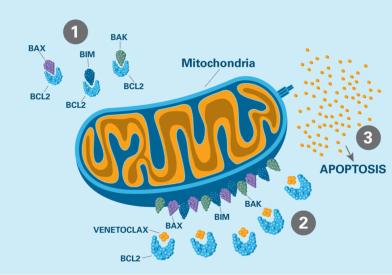
Members of our department investigate the disruption of the molecular pathways governing growth, leading to cancer. Our discoveries provide the basis for new therapies that target molecular defects in specific cancers and help to improve immune system targeting of tumors.

Structural Biology
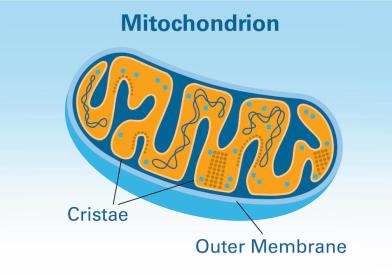
Macromolecules perform critical functions in cells. We conduct studies to determine the three-dimensional structures of cancer-associated molecules, how these structures are formed, and how structural alterations affect function.

Chemical Biology
Chemical reactions underlie core biologic processes. Chemists in our department study these reactions, synthesize bioactive molecules, and develop small molecule inhibitors for cancer therapy.

Systems Biology
Interactions of molecular and cellular components give rise to emergent behaviors of complex biological systems. We use modeling to discover biological processes that when perturbed lead to cancer.

Cancer and the Nervous System
The brain consists of multiple cell types, and the determination of the particular cellular and molecular composition of individual brain tumors is key to developing effective therapies.

Energy Homeostasis and Metabolism
The regulation of energy intake and utilization at the cellular and organismic level is of profound importance to human health. We use a combination of biochemical, genetic, and computational techniques to define key molecules and metabolic pathways.
Publications
View publications by members of the Department of Cancer Biology.
Contact Cancer Biology
If you are interested in learning more about our research, please email Priya Prakash Budde, PhD.
Support Our Cancer Research
To learn more about the many ways you can support Dana-Farber's research initiatives with a philanthropic gift, please contact Rebecca Shortle at 617-632-4215 or rebecca_shortle@dfci.harvard.edu.