Dana-Farber and Brigham and Women's researchers laud FDA approval of CAR T-cell therapy for non-Hodgkin Lymphoma
Dana-Farber/Brigham and Women's one of the only centers certified to offer new approach
Following a successful clinical trial involving Dana-Farber Cancer Institute and Brigham and Women's Hospital, the first chimeric antigen receptor (CAR) T-cell therapy for adult cancers was approved by the Food and Drug Administration (FDA) today. Dana-Farber/Brigham and Women's Cancer Center, the only facility in the northeast to be part of the clinical trial, is one of a few locations certified to offer this new therapy nationwide.
The drug, known as Yescarta (axicabtagene ciloleucel), was developed by Kite Pharma and can now be used to treat adults with refractory aggressive B cell non-Hodgkin lymphoma (NHL). The FDA ruling is based on the results of a clinical trial that showed the therapy to be safe and effective. On this nationwide trial, 101 patients received Yescarta and 82 percent of patients responded to the treatment, with 54 percent of patients having a complete response to therapy, according to results presented in June at the Lugano International Conference on Malignant Lymphoma. Thirty-six percent of patients remain in complete remission six months after treatment.
"Treating patients with CAR T-cells has been one of my most exciting professional experiences, and the FDA approval of this therapy offers hope and optimism to a subset of patients whose other treatments have failed them," says Caron A. Jacobson, MD, MMsc, Medical Director of the Immune Effector Cell Therapy program at Dana-Farber/Brigham and Women's Cancer Center. "It is extremely rewarding to be able to offer a new therapy to patients who had virtually no other options just 12 to 24 months ago."
Over the past couple of years, Jacobson and her team have been testing Yescarta in a clinical trial. The patients on this study met the following qualifications:
Diagnosed with a highly refractory fast-growing B cell non-Hodgkin lymphoma -- called diffuse large B cell lymphoma, primary mediastinal B cell lymphoma, or transformed follicular lymphoma
Did not experience a positive response to past treatments or experienced disease progression following their last chemotherapy regimen
"This therapy requires just a one-time infusion for patients, and the results are evident within one month," Jacobson said. "It is our goal as clinicians to help patients and improve their quality of life. Seeing these patients return to work, their families, and their livelihoods so quickly is an important reminder of how far we have come. It is also inspiration for the work we still need to do."
The approval follows the FDA's recent first-ever approval of CAR T-cell therapy for the treatment of some pediatric and young adult patients with B-cell acute lymphoblastic leukemia (ALL) in August.
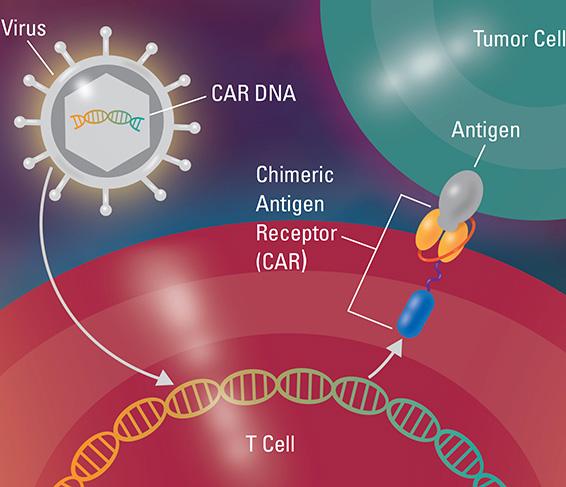
CAR T-cell therapy, like all forms of cancer immunotherapy, seeks to sharpen and strengthen the immune system's inherent cancer-fighting powers. It involves giving patients modified versions of their own immune system T-cells — white blood cells that help protect the body from disease.
To convert normal T-cells into CAR T-cells, technicians first extract T-cells from a patient's blood and genetically engineer them in a lab to produce proteins on their surface called chimeric antigen receptors, or CARs. The CARs serve a dual purpose: to enable the T-cells to latch onto specific tumor cell proteins called antigens, and to signal the T-cells to kill those tumor cells. The newly minted CAR T-cells grow in a lab until they number in the hundreds of millions, and are then infused into the patient. If successful, the CAR T-cells will continue to reproduce in the patient's body, and serve as an effective fighting force against cancer cells.
The initial clinical trials of CAR T-cell therapy have involved pediatric and adult patients with blood-based cancers such as leukemia, lymphoma, and multiple myeloma. Based on the therapy's success so far, CAR T-cell therapy trials are now opening for certain types of solid tumors as well.
"The successful development of CAR T-cells as a therapy for cancer, on the heels of the success of immune checkpoint blockade drugs, is a testament to the progress we have made in understanding how our immune system is regulated and how cancer evades the immune system," Jacobson said. "It is a perfect example of how basic science research can fuel clinical progress. Now we need to take what we can from the clinic back to the laboratory to make this therapy even better."
Media Contacts
If you are a journalist and have a question about this story, please call 617-632-4090 and ask to speak to a member of the media team, or email media@dfci.harvard.edu.
The Media Team cannot respond to patient inquiries. For more information, please see Contact Us.