Eric Fischer, PhD
Contact Information
Office Phone Number
On This Page
Research
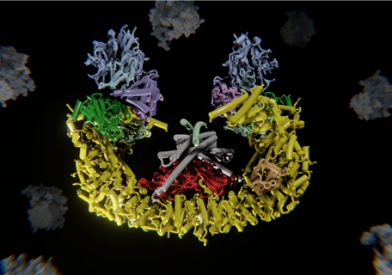
The Ubiquitin Proteasome System (UPS)
The Ubiquitin Proteasome System (UPS) is involved in virtually any cellular process and frequently implicated in human pathologies. Ubiquitin, through the action of a three-enzyme cascade (E1, E2 and E3), becomes attached to substrate proteins. The posttranslational modification with ubiquitin can serve a multitude of functions depending on the type and length of the ubiquitin chain attached to the substrate, including the control of protein abundance via proteasomal degradation. The human genome encodes for more than 600 E3 ligases, which confer specificity in the ubiquitin signaling cascade. While the process of ubiquitin transfer is well understood, the biological function and molecular mechanisms of the majority of ubiquitin ligases remain obscure.
We combine structural biology, cell biology and biochemical reconstitutions to address the molecular workings of these multi-component ubiquitin ligases. In particular, we are interested in protein complexes and pathways that contribute to the control of gene expression and are frequently associated with human disease and cancer. Intimate understanding of the structure allows us to dissect the complex mechanisms that underlie function and regulation of such molecules and to probe their biology in a cellular context. We seek to leverage our molecular understanding to propose and test new avenues of therapeutic intervention.
We are also interested in small molecules targeting the UPS such as the anti-cancer therapeutics thalidomide and derivatives. As before, we utilize a broad toolset of structural biology, proteomics, cell biology and biochemical reconstitutions to elucidate their activities and precise mode of action.